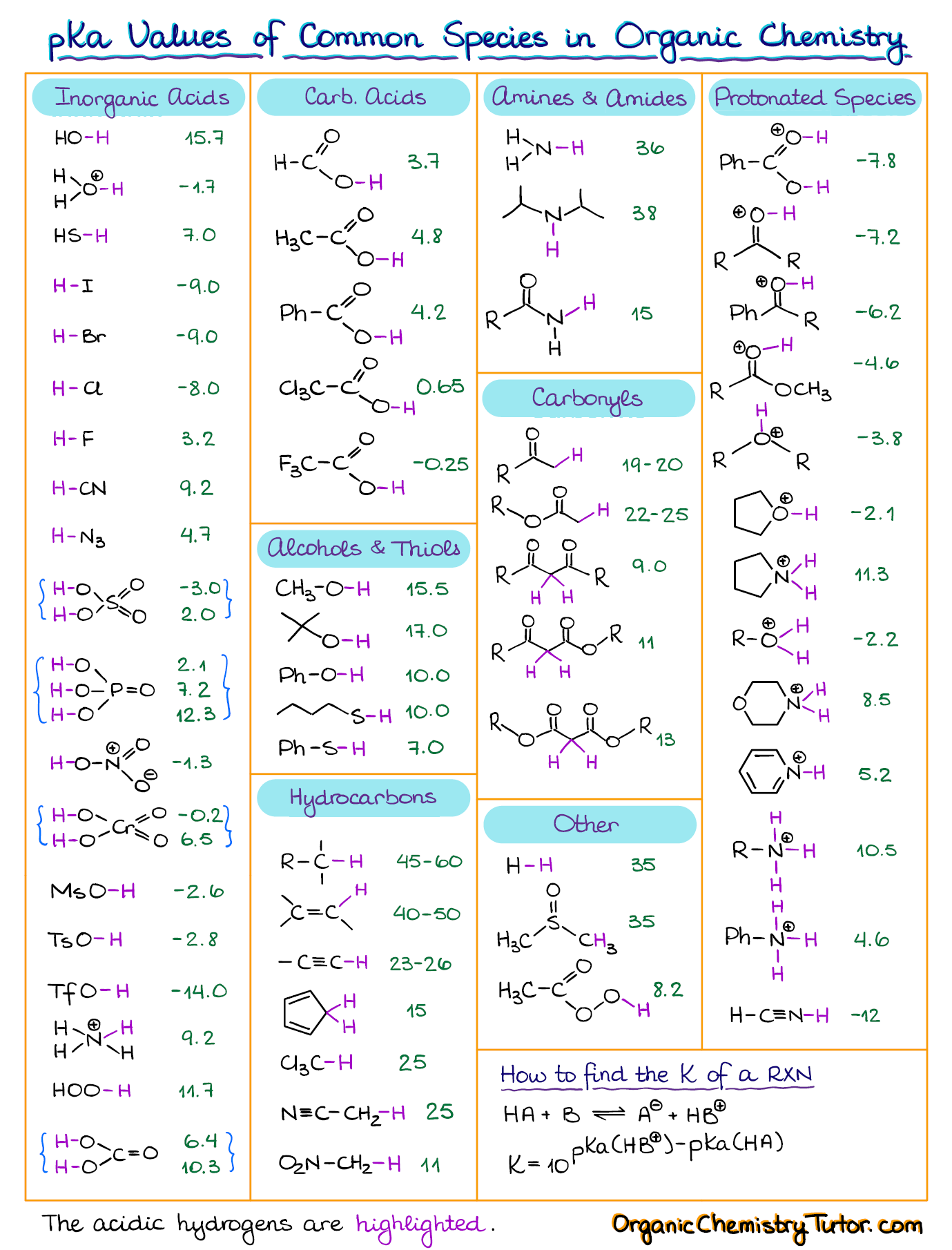
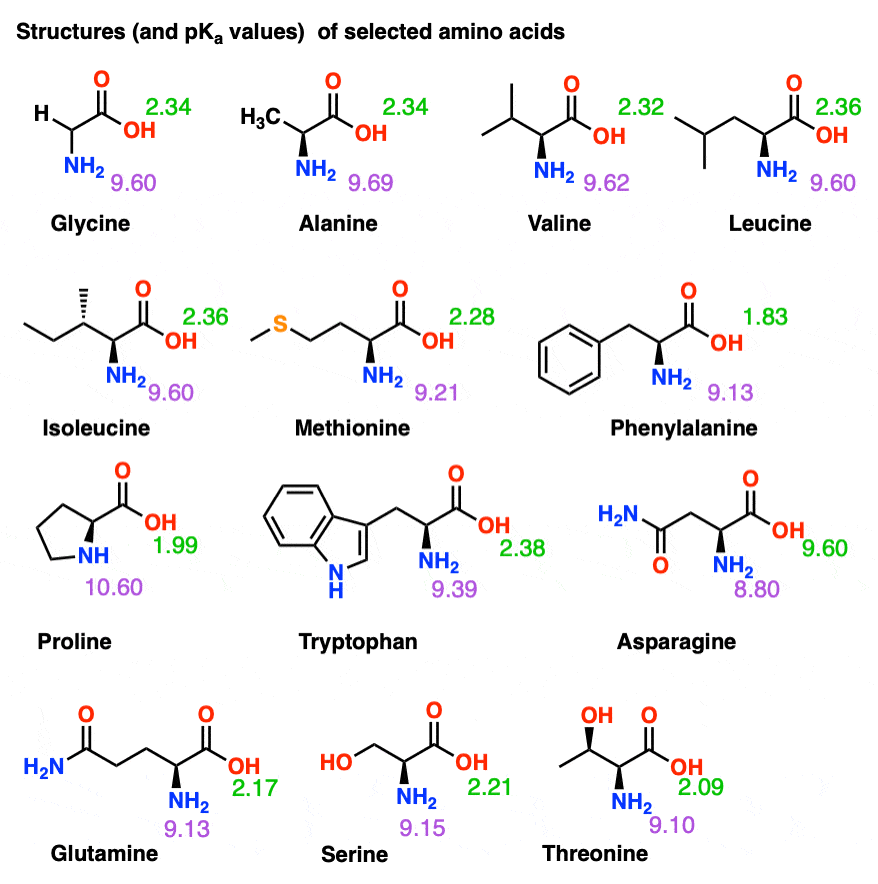
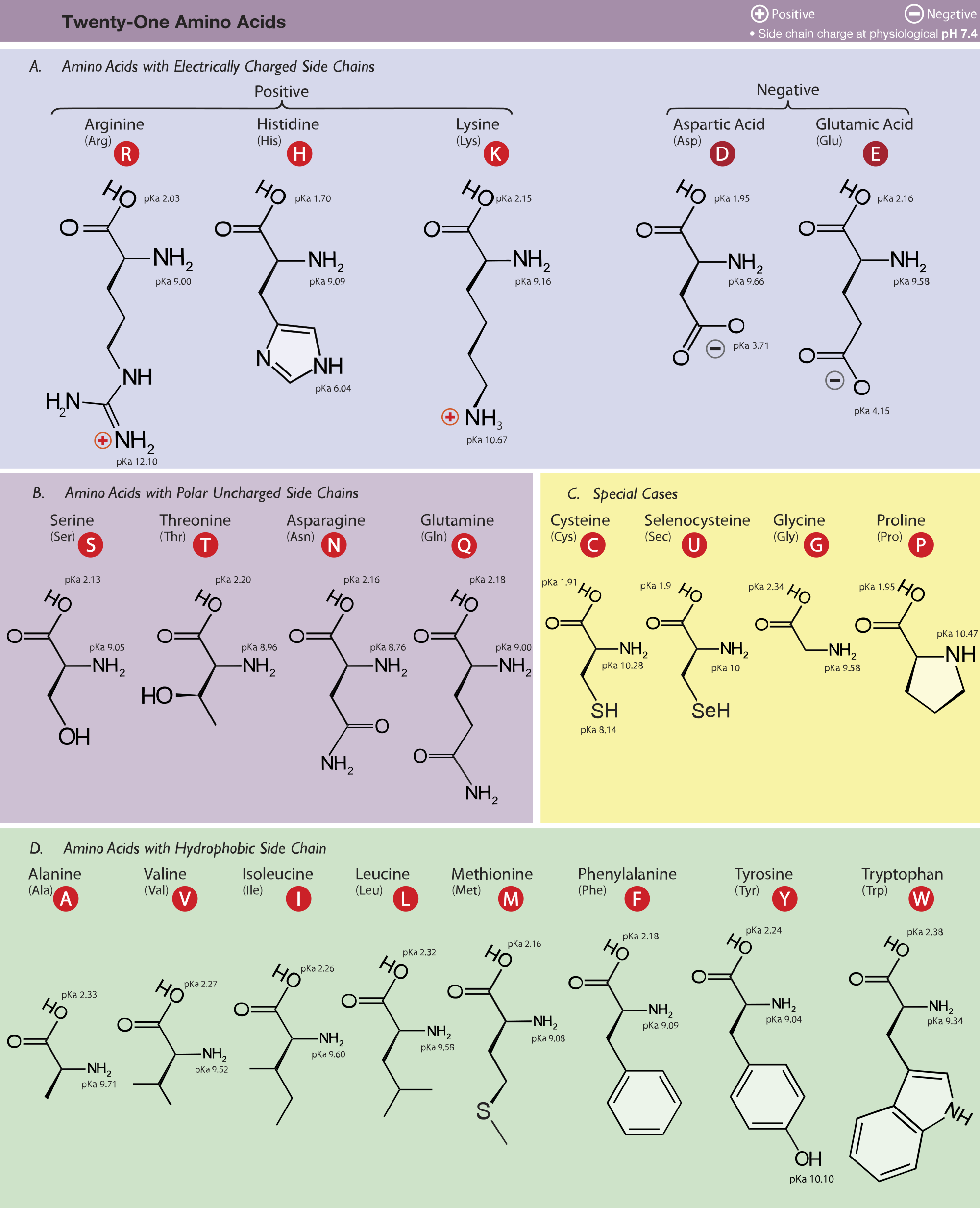
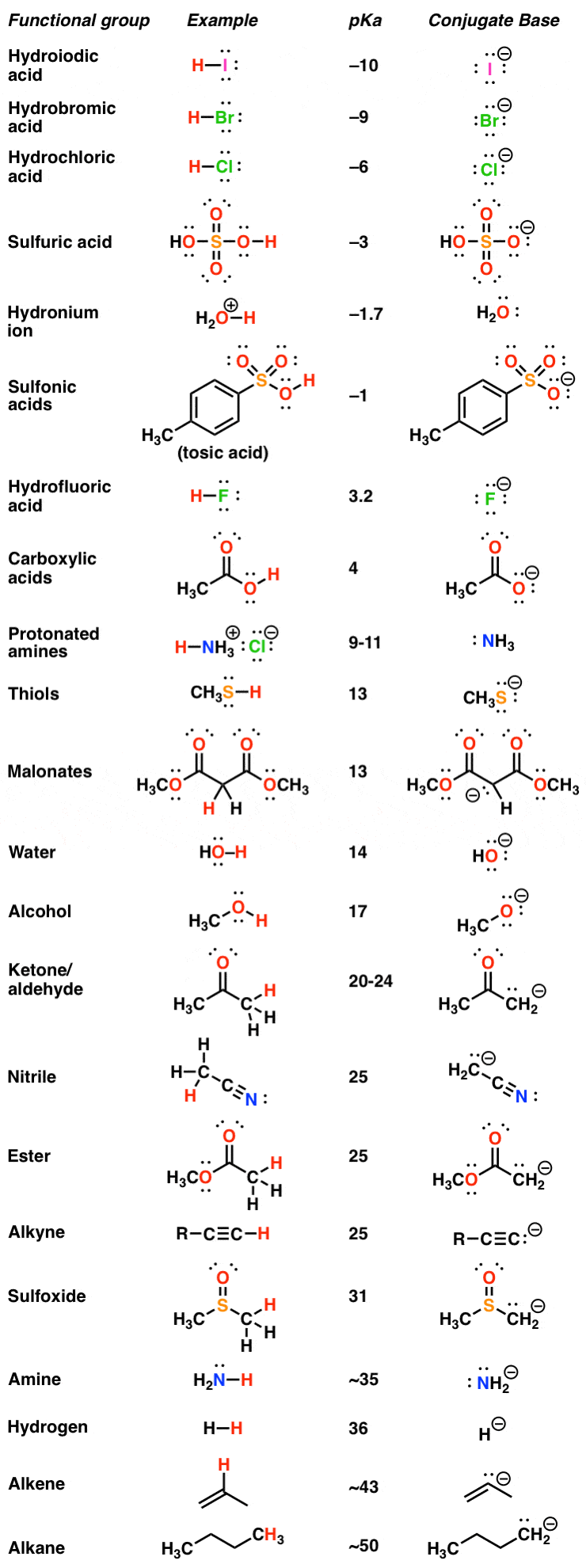
Pka Chart Amino Acids
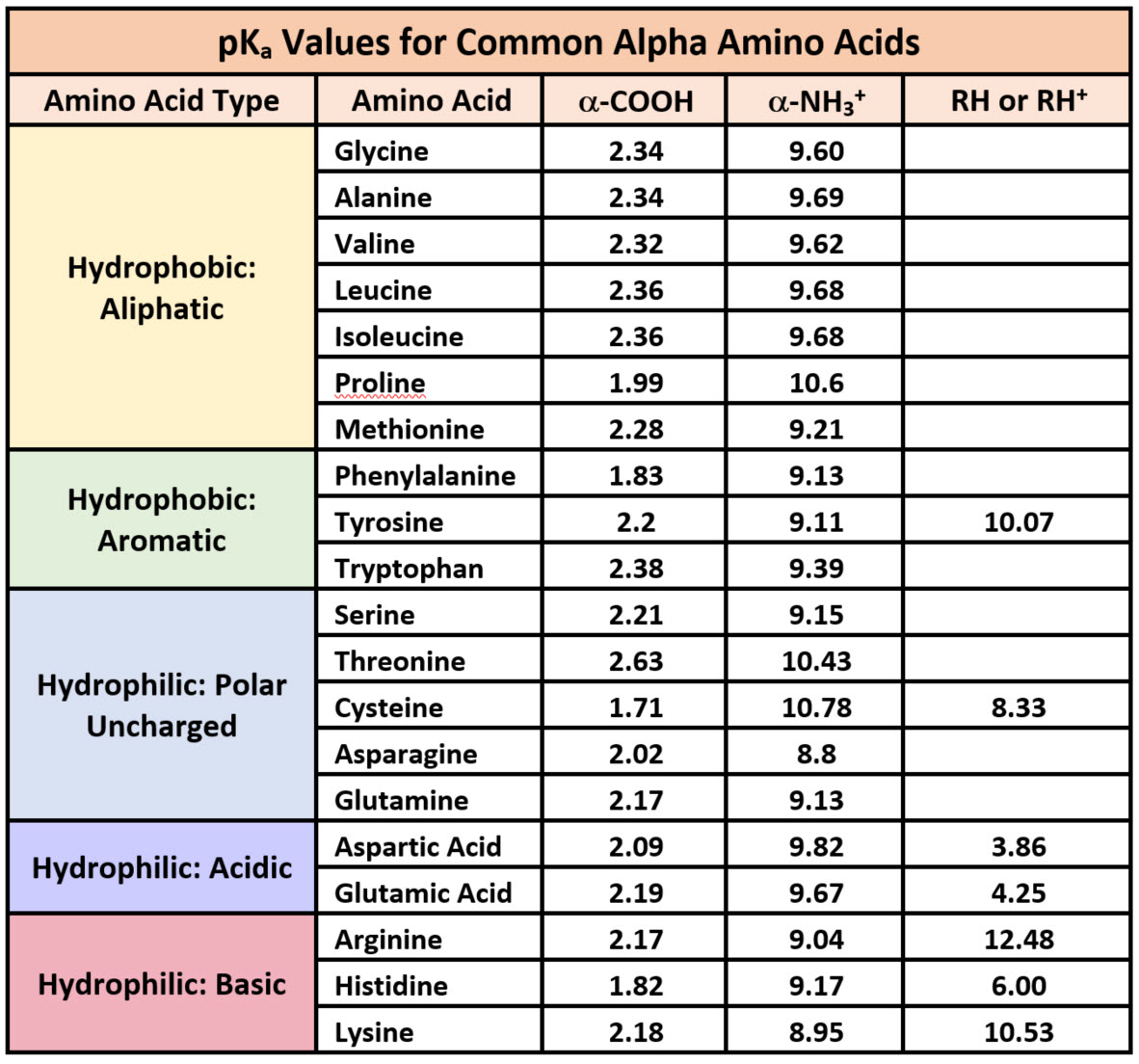
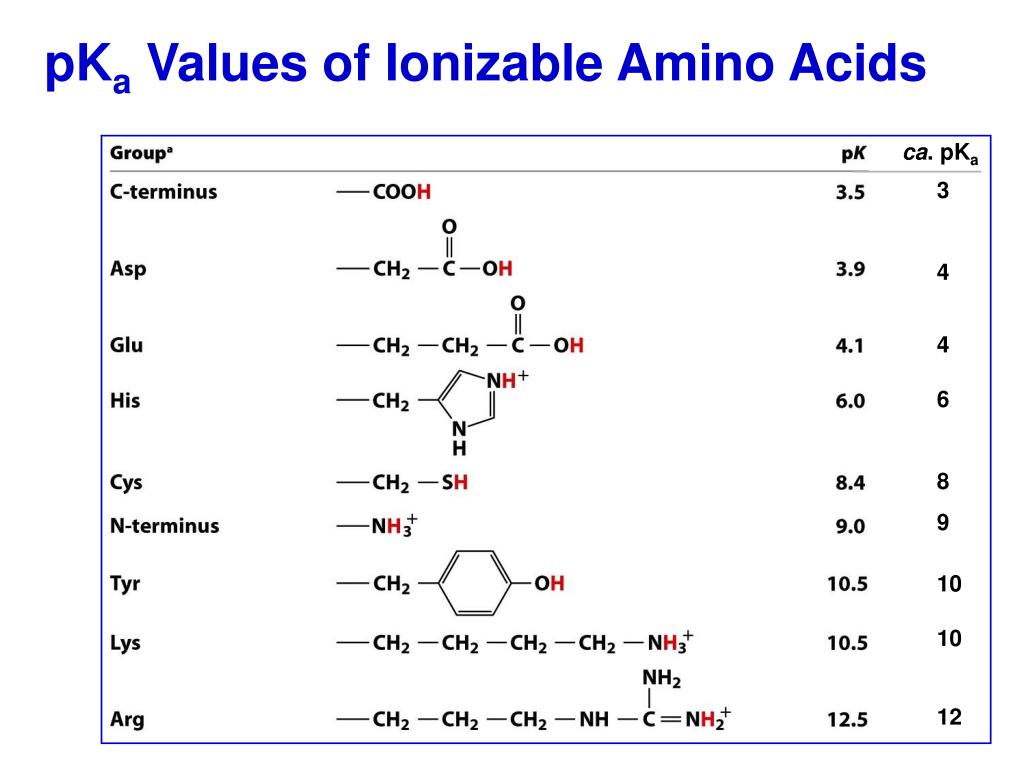
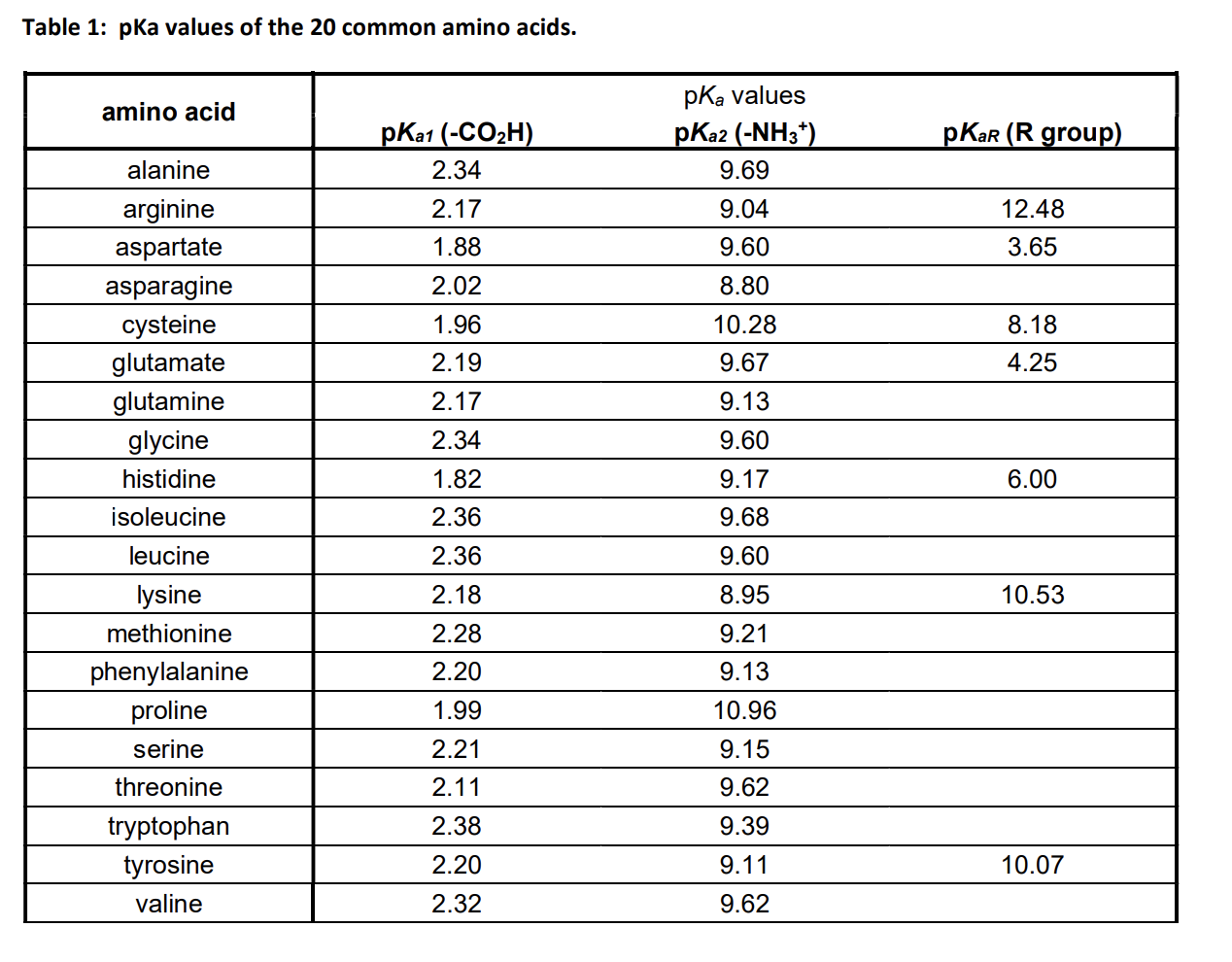
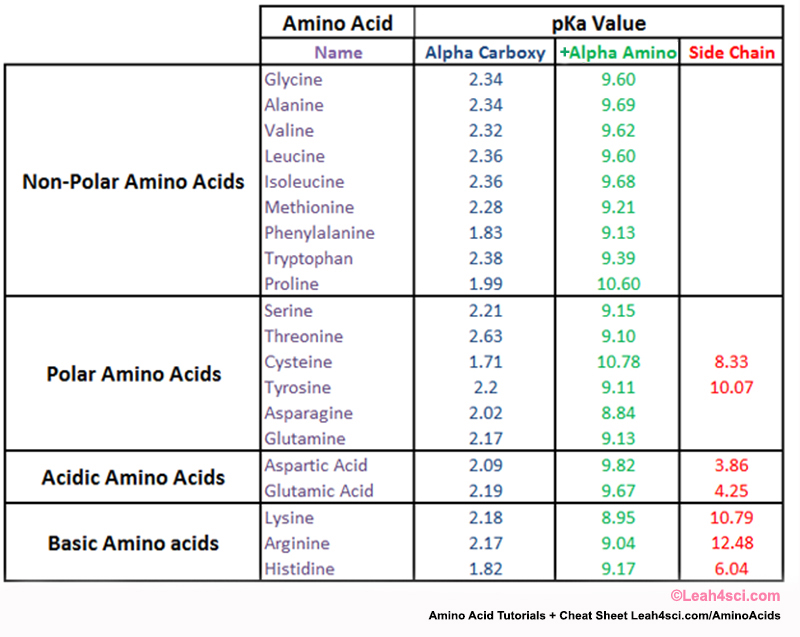
Pka Chart Amino Acids - Amino acid pka c pka n pka r pi; Web pka and electrical properties of amino acids. Web alcohols and oxygen acids 10, 11 quinoxaline 27 amino acids 12 special nitrogen compounds 28. The isoelectric points range from 5.5 to 6.2. Web 20 amino acids and their functions, structures, names, properties, classifications. Amino acids have −cooh − cooh group that is acidic with pk a 2. Web basic molecules gain protons and acidic molecules donate protons. Web most biochemistry courses will require you to know the following: Web kainoid synthases are key enzymes in the biosynthesis of kainoids. Web you should be able to classify all the amino acids by polarity, charge, aliphatic vs aromatic, and probably learn the structures and functional groups of the special amino acids (for. Amino acids have −cooh − cooh group that is acidic with pk a 2. Web pka is an acid dissociation constant used to describe the acidity of a particular molecule. Does one of these forms dominate the equilibrium? Amino acids, peptides and proteins. It represents the negative logarithm of the acid. Its value is directly related to the structure of the given compound. Amino acid pka c pka n pka r pi; At neutral ph the amino group is protonated, and the carboxyl. Web pka and electrical properties of amino acids. Web alcohols and oxygen acids 10, 11 quinoxaline 27 amino acids 12 special nitrogen compounds 28. Web this is why the carboxylic acid groups of amino acids have a lower pka value of around 2, while acetic acid has a pka value of 4.76. Web two forms of an amino acid, related by proton transfer. It represents the negative logarithm of the acid. There are 22 amino acids that are found in proteins and of these,. Web the r group for each of the amino acids will differ in structure, electrical charge, and polarity. Web the pka is a measure of the strength of an acid, i.e., the lower the pk a stronger the acid. Web most biochemistry courses will require you to know the following: Web this is why the carboxylic acid groups of amino. There are 22 amino acids that are found in proteins and of these, only 20 are specified by the universal genetic code. Web this is why the carboxylic acid groups of amino acids have a lower pka value of around 2, while acetic acid has a pka value of 4.76. Certain functional groups give away. Web you should be able. Certain functional groups give away. Web pka is an acid dissociation constant used to describe the acidity of a particular molecule. Web most biochemistry courses will require you to know the following: At neutral ph the amino group is protonated, and the carboxyl. Web amino acid pka and pi values. In organic chemistry, pka is a measure of the acidity or basicity of a compound. Web alcohols and oxygen acids 10, 11 quinoxaline 27 amino acids 12 special nitrogen compounds 28. Web two forms of an amino acid, related by proton transfer. Cyanoamines 16 amidoximes 28 anilines 17, 18, 20 thiols 29. Its value is directly related to the structure. Its value is directly related to the structure of the given compound. Web pka is an acid dissociation constant used to describe the acidity of a particular molecule. There are 22 amino acids that are found in proteins and of these, only 20 are specified by the universal genetic code. The pka of the acid is near 5, and the. Web most biochemistry courses will require you to know the following: Web the pka is a measure of the strength of an acid, i.e., the lower the pk a stronger the acid. Amino acid pka and pi values Certain functional groups give away. It represents the negative logarithm of the acid. Does one of these forms dominate the equilibrium? At neutral ph the amino group is protonated, and the carboxyl. Glycine alanine valine leucine isoleucine proline serine threonine cysteine methionine asparagine. Its value is directly related to the structure of the given compound. For the 13 amino acids with a neutral side. Table of pka and pi values. The isoelectric points range from 5.5 to 6.2. Web the pka is a measure of the strength of an acid, i.e., the lower the pk a stronger the acid. Does one of these forms dominate the equilibrium? Web 20 amino acids and their functions, structures, names, properties, classifications. The isoelectric points range from 5.5 to 6.2. Amino acids have −cooh − cooh group that is acidic with pk a 2. Does one of these forms dominate the equilibrium? Amino acid pka and pi values Web 20 amino acids and their functions, structures, names, properties, classifications. Web 20 amino acids and their functions, structures, names, properties, classifications. Table of pka and pi values. Web basic molecules gain protons and acidic molecules donate protons. Web the r group for each of the amino acids will differ in structure, electrical charge, and polarity. Web kainoid synthases are key enzymes in the biosynthesis of kainoids. Its value is directly related to the structure of the given compound. Web most biochemistry courses will require you to know the following: The pka of the acid is near 5, and the pka of the. Web this is why the carboxylic acid groups of amino acids have a lower pka value of around 2, while acetic acid has a pka value of 4.76. Amino acid pka and pi values Amino acid pka c pka n pka r pi; Refer to the charts and structures below to explore amino acid properties,. If a molecule is a base or an acid, depends on their functional groups. Web two forms of an amino acid, related by proton transfer. Web pka is an acid dissociation constant used to describe the acidity of a particular molecule. Web amino acid pka and pi values.3.1 Amino Acids and Peptides Biology LibreTexts
Amino Acid Pka Chart A Visual Reference of Charts Chart Master
Amino Acids Pka Chart
Solved Table 1 pKa values of the 20 common amino acids.
Amino Acid Charge in Zwitterions and Isoelectric Point MCAT Tutorial
AcidBase Equilibrium Part 1 How to Use the pKa Table — Organic
Isoelectric Points of Amino Acids (and How To Calculate Them) Master
Amino Acid Study Guide Structure and Function Albert.io
AcidBase Reactions Introducing Ka and pKa Master Organic Chemistry
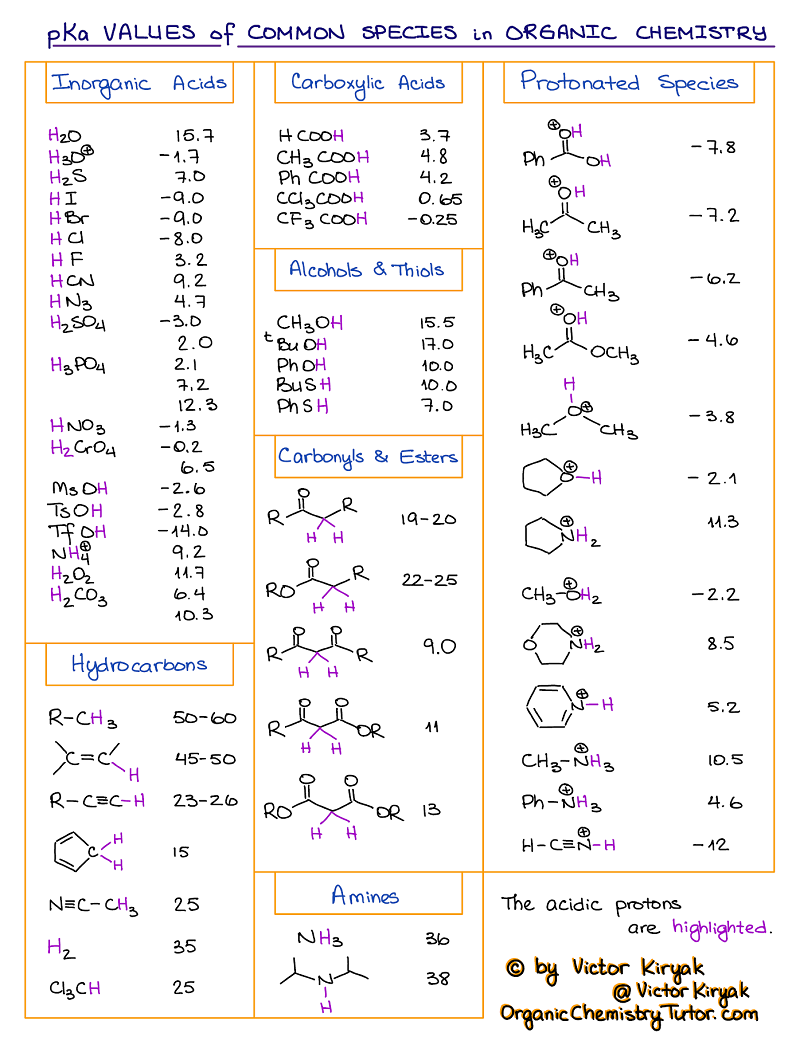
pKa Table and How to Use It — Organic Chemistry Tutor
Web The Pka Is A Measure Of The Strength Of An Acid, I.e., The Lower The Pk A Stronger The Acid.
It Represents The Negative Logarithm Of The Acid.
Web Pka And Electrical Properties Of Amino Acids.
For The 13 Amino Acids With A Neutral Side.
Related Post: