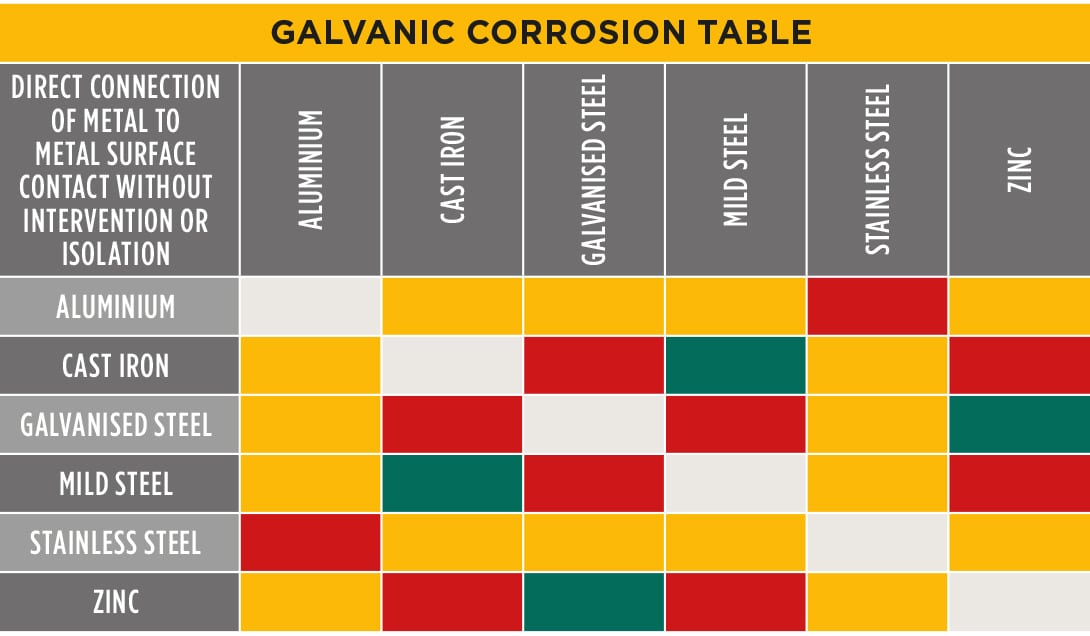
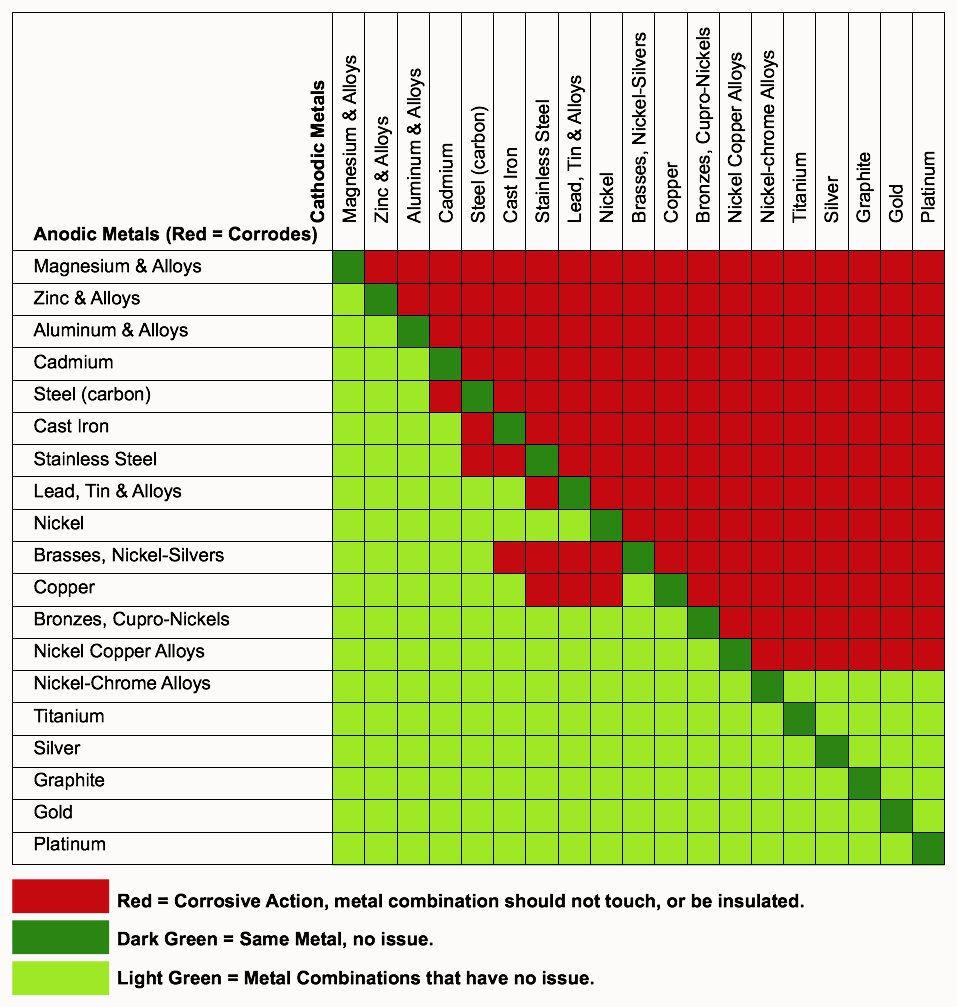
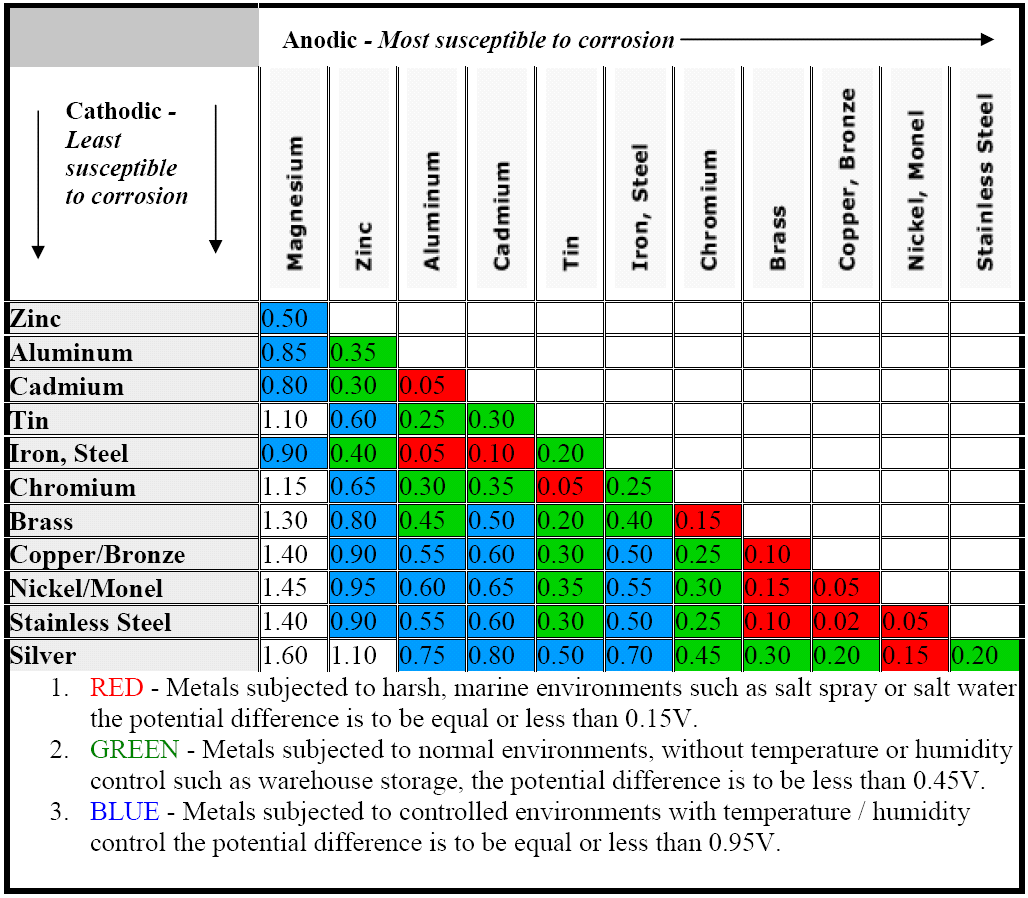
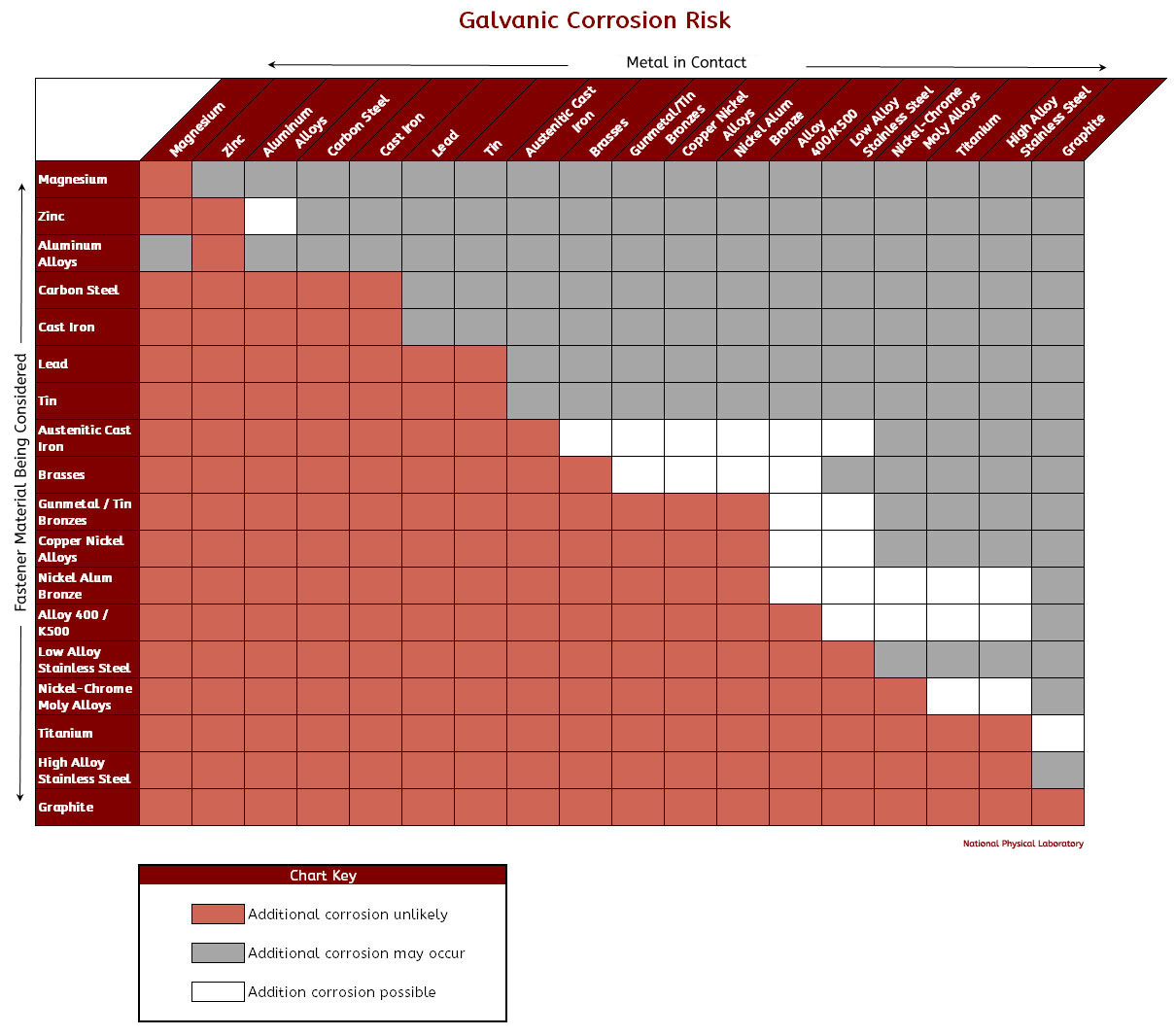
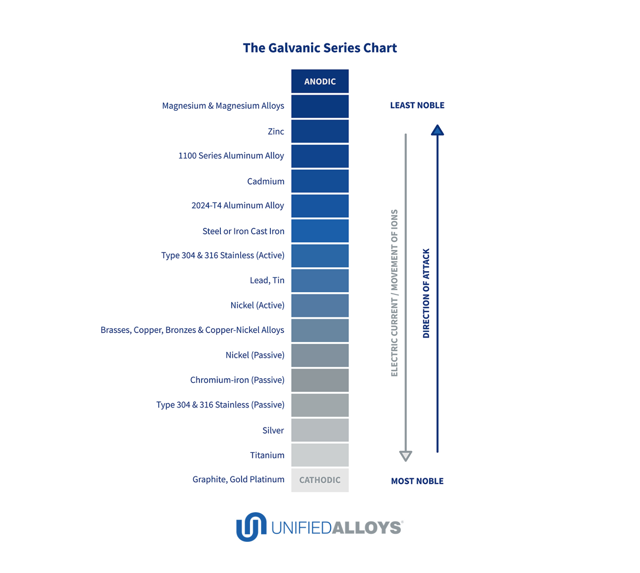
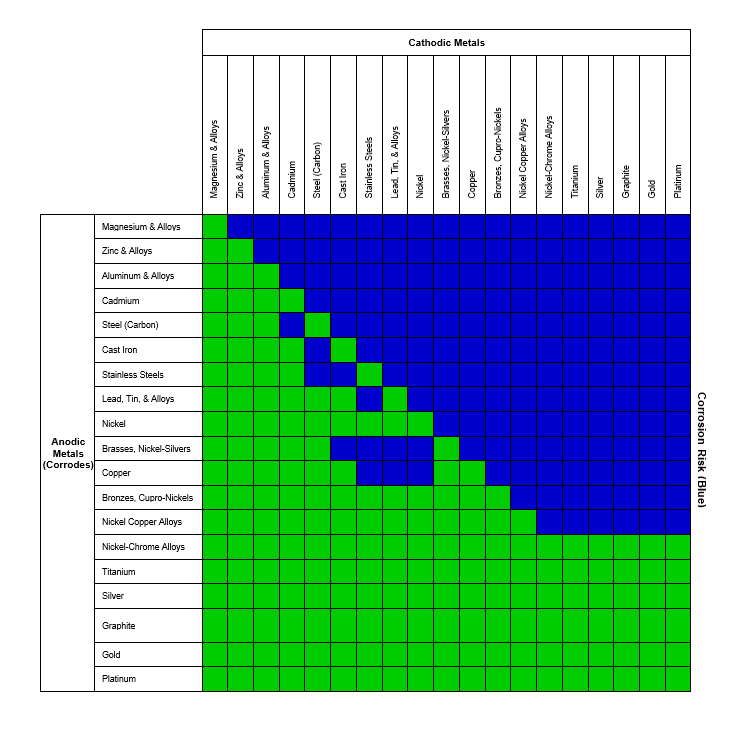
Galvanic Coupling Chart
Galvanic Coupling Chart - The small surface area of the active bolts results in an undesirable galvanic couple and they are exhibiting an accelerated corrosion rate. Hydrogen evolution (acids) 2h + + 2e ~ h2. Web the galvanic series compatibility of different metals can be assessed, relative to the potential for galvanic corrosion, with the use of charts depicting the galvanic (or electromotive force) series in different environments. Web there are two primary types of galvanic cells that cause corrosion: You can also learn more about overcoming potentially compatibility issues between metals. Web below, we give a brief overview of galvanic corrosion and provide a galvanic corrosion chart to help fabricators and machinists avoid using the wrong metal combinations. Web figure 3a shows the galvanic corrosion of carbon steel bolts used to secure a stainless steel structural railing support on a bridge. Galvanic series / galvanic table. Web galvanic corrosion (also called bimetallic corrosion or dissimilar metal corrosion) is an electrochemical process in which one metal corrodes preferentially when it is in electrical contact with another, in the presence of an electrolyte. Web the galvanic corrosion table ranks metals from the most “active” to the least active. Web the galvanic series chart below shows metals and their electrochemical voltage range (relative activity in flowing sea water). Hydrogen evolution (acids) 2h + + 2e ~ h2. A typical rule of thumb is that voltage differences of 0.2 volts or more suggest a galvanic corrosion risk. Web galvanic corrosion (also called bimetallic corrosion or dissimilar metal corrosion) is an electrochemical process in which one metal corrodes preferentially when it is in electrical contact with another, in the presence of an electrolyte. ~ fe 2+ + 2e) and there are several possible cathodic reactions: Galvanic series / galvanic table. The list begins with the more active (anodic) metal and proceeds down. The finishing and plating selected facilitate the dissimilar materials being in contact and protect the base materials from corrosion. A classification of the different metals and alloys according to this measured potential (see chart below). Web to minimize galvanic corrosion, select fasteners based on their material compatibility with the substrates. Web view this chart of galvanic compatibility. Web however, you can completely avoid galvanic corrosion by choosing matching metal anchors. Web figure 3a shows the galvanic corrosion of carbon steel bolts used to secure a stainless steel structural railing support on a bridge. The corroded area was machined out and rebuilt with alloy 625 filler metal which is cathodic to. The most active metals in the galvanic corrosion chart, like aluminum, zinc, or magnesium, are more likely to corrode when connected to. In this article, we'll look at an example to illustrate the use of the galvanic table. This form of corrosion has the potential to attack junctions of metals, or regions where one construction ~ fe 2+ + 2e). So, for example, choosing zinc on zinc would have the lowest risk for corrosion. The small surface area of the active bolts results in an undesirable galvanic couple and they are exhibiting an accelerated corrosion rate. This form of corrosion has the potential to attack junctions of metals, or regions where one construction Web figure 3a shows the galvanic corrosion. You can also learn more about overcoming potentially compatibility issues between metals. Web view this chart of galvanic compatibility. Metals listed on the top of the chart (anodic) will corrode faster than the metals on the bottom of the chart (cathodic). Hydrogen evolution (acids) 2h + + 2e ~ h2. Web galvanic corrosion is a localised mechanism by which metals. Web the galvanic series chart below shows metals and their electrochemical voltage range (relative activity in flowing sea water). Web to minimize galvanic corrosion, select fasteners based on their material compatibility with the substrates. The closer together the material are on the chart to the right, the less galvanic action will occur. Web often when design requires that dissimilar metals. Web figure 3a shows the galvanic corrosion of carbon steel bolts used to secure a stainless steel structural railing support on a bridge. Web by knowing the relationships of the metals in the series, galvanic compatibility can be determined, preventing the possible harmful effects of galvanic corrosion. In this article, we'll look at an example to illustrate the use of. Web often when design requires that dissimilar metals come in contact, the galvanic compatibility is managed by finishes and plating. The following galvanic table lists metals in the order of their relative activity in seawater environment. Web read on to find out about what it is and how to use it to analyse the compatibility of joining metals. Web there. Web often when design requires that dissimilar metals come in contact, the galvanic compatibility is managed by finishes and plating. Web there are two primary types of galvanic cells that cause corrosion: Use this chart below to better understand what metals will work best together without potential for galvanic corrosion: Web in each solution, it is possible to establish a. Web galvanic corrosion (also called bimetallic corrosion or dissimilar metal corrosion) is an electrochemical process in which one metal corrodes preferentially when it is in electrical contact with another, in the presence of an electrolyte. In this article, we'll look at an example to illustrate the use of the galvanic table. Web by knowing the relationships of the metals in. The closer together the material are on the chart to the right, the less galvanic action will occur. A classification of the different metals and alloys according to this measured potential (see chart below). Web galvanic corrosion undermined the keeper rings, leading to failure and leakage. Web the galvanic corrosion table ranks metals from the most “active” to the least. Web the galvanic corrosion table ranks metals from the most “active” to the least active. Web the galvanic series chart below shows metals and their electrochemical voltage range (relative activity in flowing sea water). The galvanic series indicates which dissimilar metal will tend to corrode (anode) and which dissimilar metal Web galvanic corrosion is a localised mechanism by which metals can be preferentially corroded. Hydrogen evolution (acids) 2h + + 2e ~ h2. Web below is a galvanic reaction chart for dissimilar metals. A typical rule of thumb is that voltage differences of 0.2 volts or more suggest a galvanic corrosion risk. Web there are two primary types of galvanic cells that cause corrosion: Web below, we give a brief overview of galvanic corrosion and provide a galvanic corrosion chart to help fabricators and machinists avoid using the wrong metal combinations. The corroded area was machined out and rebuilt with alloy 625 filler metal which is cathodic to the copper nickel piping. The most active metals in the galvanic corrosion chart, like aluminum, zinc, or magnesium, are more likely to corrode when connected to. So, for example, choosing zinc on zinc would have the lowest risk for corrosion. Web however, you can completely avoid galvanic corrosion by choosing matching metal anchors. Web to minimize galvanic corrosion, select fasteners based on their material compatibility with the substrates. This form of corrosion has the potential to attack junctions of metals, or regions where one construction In this article, we'll look at an example to illustrate the use of the galvanic table.Galvanic Corrosion Cable Cleats CMP Products Limited
Galvanic Action Corrosion Prevention Architect's Blog
Galvanic Chart FINE METAL ROOF TECH
Galvanic Corrosion A Guide for Architects (with a Galvanic Series Chart)
Chemical Resistance Chart For Metals
Galvanic Corrosion Chart
Galvanic Corrosion Chart Metals
Stainless Steel Galvanic Corrosion Chart
Galvanic Corrosion Chart Metals
Galvanic Corrosion Chart Industrial Metal Service
Web Galvanic Corrosion (Also Called Bimetallic Corrosion Or Dissimilar Metal Corrosion) Is An Electrochemical Process In Which One Metal Corrodes Preferentially When It Is In Electrical Contact With Another, In The Presence Of An Electrolyte.
The Closer Together The Material Are On The Chart To The Right, The Less Galvanic Action Will Occur.
Web By Knowing The Relationships Of The Metals In The Series, Galvanic Compatibility Can Be Determined, Preventing The Possible Harmful Effects Of Galvanic Corrosion.
The List Begins With The More Active (Anodic) Metal And Proceeds Down.
Related Post: