Gadavist Dose Chart
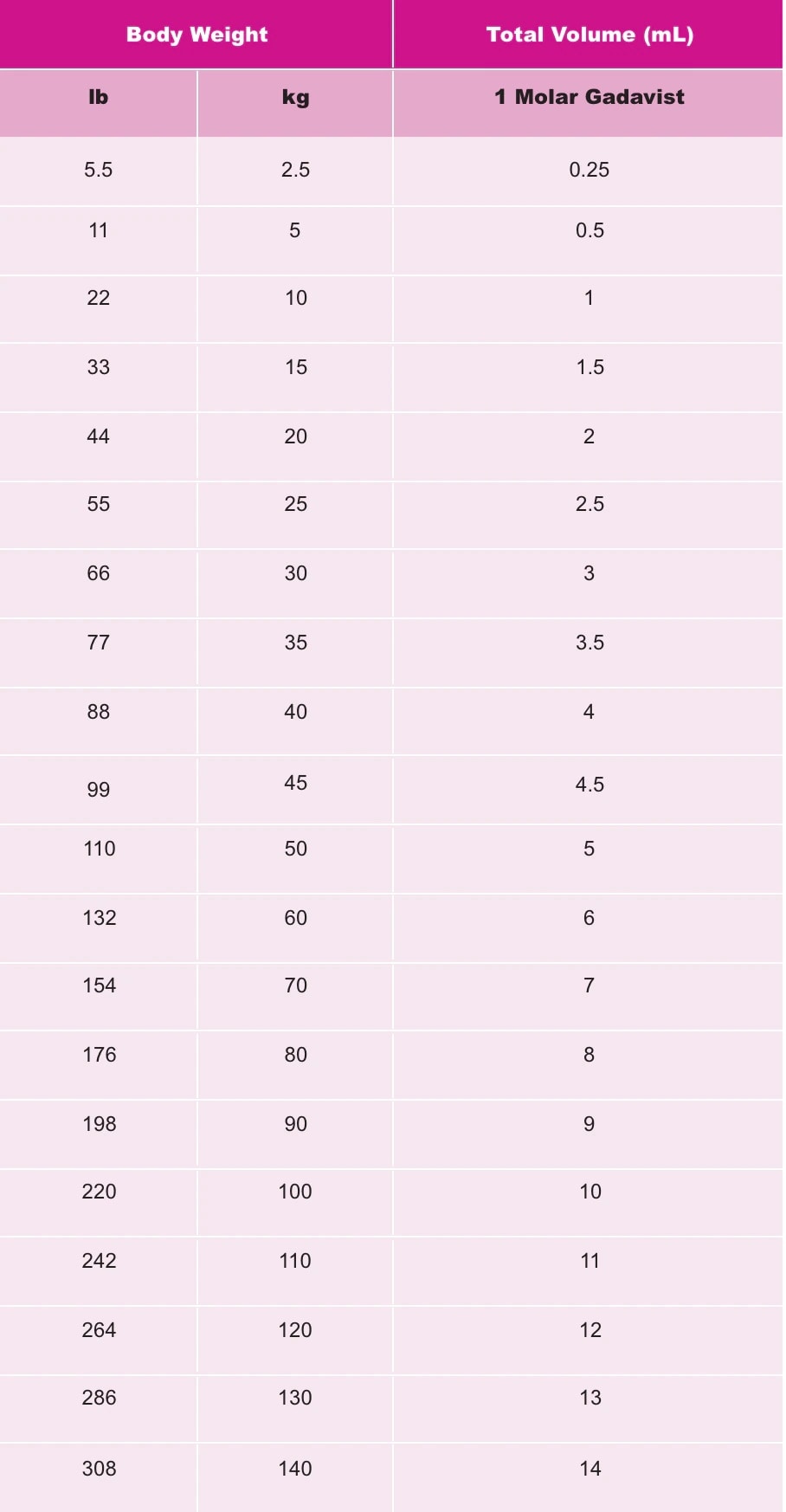
Gadavist Dose Chart - Refer to table 1 to determine the volume to be. Web the recommended dose of gadavist for adult and pediatric patients (including term neonates) is 0.1 ml/kg body weight mmol/kg). Use the table in section 2.1. Administer intravenously as a bolus, manually or by power injector. Gadavist, like other gbcas, is injected into your vein and used with a magnetic resonance imaging (mri) scanner. 0.1 ml/kg) administered intravenously at approximately 2 ml/sec. 1mmol/ml (ie, 604.72mg/ml) mri contrast. Web the recommended dose of gadavist for adults and children of all ages (including term neonates) is 0.1 ml/kg body weight (0.1 mmol/kg) to be administered as an intravenous Web at equivalent doses, the amount of gadolinium that stays in the body is different for different gadolinium medicines. Usual adult dose for cns magnetic resonance imaging: Refer to table 1 to determine the volume to be administered. The recommended dose of gadavist for adult and pediatric patients (including term neonates) is 0.1 ml/kg body weight mmol/kg). Refer to table 1 to determine the volume to be. Web the recommended dose of gadavist for adults and children of all ages (including term neonates) is 0.1 ml/kg body weight (0.1 mmol/kg) to be administered as an intravenous Web • the recommended dose for adult and pediatric patients aged 2 years and older is 0.05 mmol/kg actual body weight (equivalent to. Refer to table 1 to determine the volume to be administered. Gadolinium stays in the body more after omniscan or optimark than after eovist, magnevist, or multihance. Administer intravenously as a bolus, manually or by power injector. Recommended dosage with gadovist® 1.0 depends on the indication1. Web at equivalent doses, the amount of gadolinium that stays in the body is different for different gadolinium medicines. (0.1 mmol/kg body weight) is sufficient to answer the clinical questions.1. Maximum recommended dose at uw. To detect and visualize areas with disrupted blood brain barrier and/or abnormal vascularity of the central nervous system in adult and pediatric patients including term neonates. Web • the recommended dose for adult and pediatric patients aged 2 years and older is 0.05 mmol/kg. Web • the recommended dose for adult and pediatric patients aged 2 years and older is 0.05 mmol/kg actual body weight (equivalent to. Note maximum dose of eovist is 0.05mmol/kg. To detect and visualize areas with disrupted blood brain barrier and/or abnormal vascularity of the central nervous system in adult and pediatric patients including term neonates. Web the recommended dose. Gadolinium stays in the body more after omniscan or optimark than after eovist, magnevist, or multihance. Web screen patients for acute kidney injury and other conditions that may reduce renal function. 0.1 ml/kg) administered intravenously at approximately 2 ml/sec. Note maximum dose of eovist is 0.05mmol/kg. Administer intravenously as a bolus, manually or by power injector. Refer to table 1 to determine the volume to. Usual adult dose for cns magnetic resonance imaging: Use the table in section 2.1. Web at equivalent doses, the amount of gadolinium that stays in the body is different for different gadolinium medicines. The recommended dose of gadavist for adult and pediatric patients (including term neonates) is 0.1 ml/kg body weight. Web screen patients for acute kidney injury and other conditions that may reduce renal function. Generally, a single injection of 0.1 ml/kg body weight. Gadolinium stays in the body more after omniscan or optimark than after eovist, magnevist, or multihance. To detect and visualize areas with disrupted blood brain barrier and/or abnormal vascularity of the central nervous system in adult. Administer manually or by power injector at a flow rate of approximately 2 ml/second. To detect and visualize areas with disrupted blood brain barrier and/or abnormal vascularity of the central nervous system in adult and pediatric patients including term neonates. Volume of gadavist injection by body weight* Use the table in section 2.1. Web the recommended dose of gadavist is. Gadavist, like other gbcas, is injected into your vein and used with a magnetic resonance imaging (mri) scanner. Web gadolinium based contrast dosing charts: Administer manually or by power injector at a flow rate of approximately 2 ml/second. Web • the recommended dose for adult and pediatric patients aged 2 years and older is 0.05 mmol/kg actual body weight (equivalent. Gadolinium stays in the body the least after dotarem, gadavist, or prohance. Web dosage for gadavist recommended dose. Since receiving fda approval in 2011, gadavist ® has received 3 additional indications. Administer manually or by power injector at a flow rate of approximately 2 ml/second. Administer intravenously as a bolus, manually or by power injector. Web the recommended dose of gadavist is 0.1 ml/kg body weight (0.1 mmol/kg), administered as an intravenous bolus injection at a flow rate of approximately 2 ml/second. Web the recommended dose of gadavist for adult and pediatric patients (including term neonates) is 0.1 ml/kg body weight (0.1 mmol/kg). Usual adult dose for cns magnetic resonance imaging: 0.1 ml/kg) administered intravenously. (0.1 mmol/kg body weight) is sufficient to answer the clinical questions.1. Administer manually or by power injector at a flow rate of approximately 2 ml/second. Web the recommended dose of gadavist for adult and pediatric patients (including term neonates) is 0.1 ml/kg body weight (0.1 mmol/kg). Maximum recommended dose at uw. Gadavist, like other gbcas, is injected into your vein. Refer to table 1 to determine the volume to be administered. Package insert dose of eovist is 0.025mmol/kg. Recommended dosage with gadovist® 1.0 depends on the indication1. Includes dose adjustments, warnings and precautions. 0.1 mmol/kg (0.1 ml/kg) iv bolus. Web gadolinium based contrast dosing charts: Web the recommended dose of gadavist for adult and pediatric patients (including term neonates) is 0.1 ml/kg b ody weight (0.1 mmol/kg). (0.1 mmol/kg body weight) is sufficient to answer the clinical questions.1. Gadolinium stays in the body more after omniscan or optimark than after eovist, magnevist, or multihance. Web • the recommended dose for adult and pediatric patients aged 2 years and older is 0.05 mmol/kg actual body weight (equivalent to. Web the recommended dose of gadavist is 0.1 ml/kg body weight (0.1 mmol/kg), administered as an intravenous bolus injection at a flow rate of approximately 2 ml/second. Note maximum dose of eovist is 0.05mmol/kg. Web detailed dosage guidelines and administration information for gadavist (gadobutrol). Web the recommended dose of gadavist for adult and pediatric patients (including term neonates) is 0.1 ml/kg body weight (0.1 mmol/kg). Refer to table 1 to determine the volume to be. Use the table in section 2.1.Gadavist (gadobutrol) injection Radiology US
Gadavist Contrast Media Gadobutrol 1 mmol / mL Intravenous Injection
Gadavist (gadobutrol) injection Radiology US
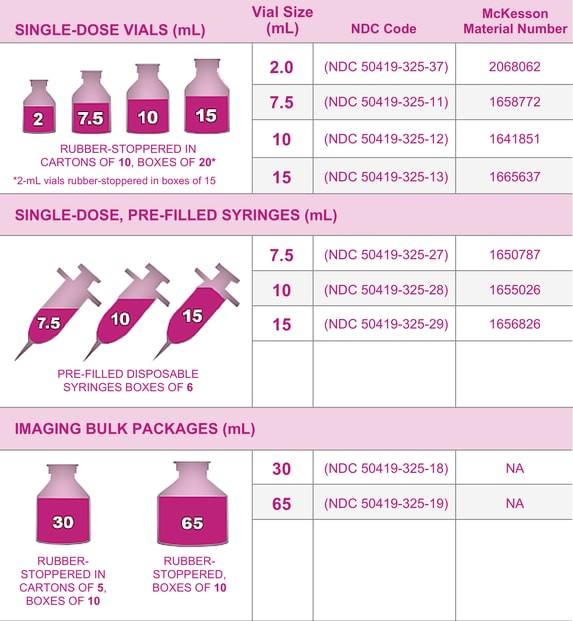
Medrad, NDC 5041932513, Bayer Gadavist (Gadobutrol) Inje... eSutures
Gadavist (gadobutrol) injection Radiology US
Clinical and biochemical characteristics of LADA patients at the time
Gadavist (gadobutrol) injection Radiology US
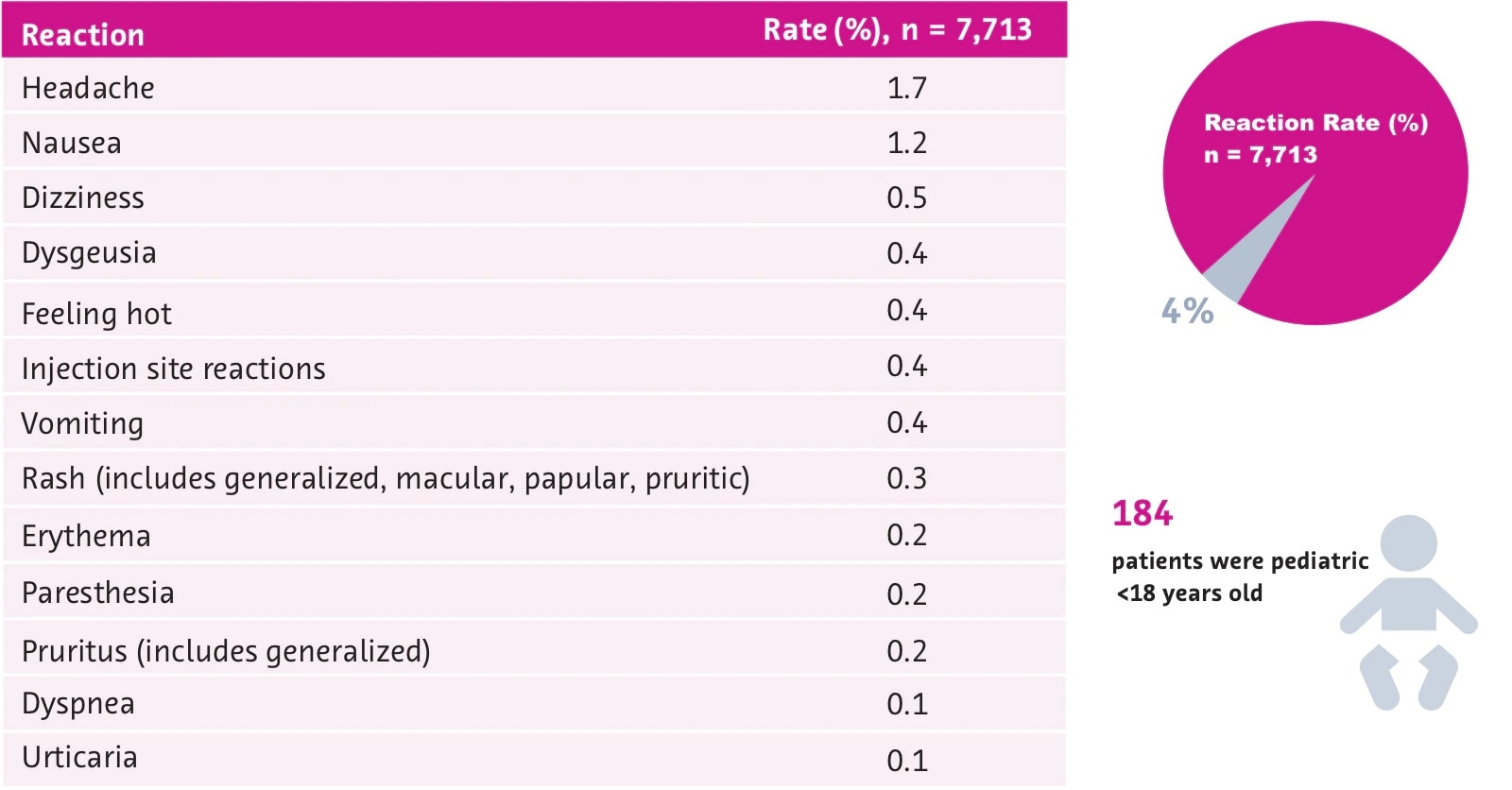
Stability of Gadolinium Based Contrast Agents (GBCAs)
Gadavist™ Gadobutrol 1 mmol / mL Injection 10 mL McKesson
Refer To Manufacturer Product Information To Determine Volume To Be Administered.
Web The Results Obtained With The Gadolinium Dose Calculator Are Intended To Serve Solely As Guidelines For Physicians And Mri Technologists And Should Be Considered As Indicative Only.
To Detect And Visualize Areas With Disrupted Blood Brain Barrier And/Or Abnormal Vascularity Of The Central Nervous System In Adult And Pediatric Patients Including Term Neonates.
Web Dosage For Gadavist Recommended Dose.
Related Post: